AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Isoelectric point calculator peptide12/18/2023  
These techniques rely on the separation of molecules based on their charge and size, and the isoelectric point can be used to predict where molecules will move in these systems. 
In addition to solubility, the isoelectric point can affect the behavior of molecules during chromatography and electrophoresis. Conversely, if the pH is above the isoelectric point, the molecule will have a net negative charge and be repelled by a negatively charged surface, also decreasing its solubility. If the pH of the solution is below the isoelectric point of the amino acid, the molecule will have a net positive charge and will be attracted to negatively charged surfaces, making it less soluble. This may affect its solubility, as charged molecules tend to dissolve more readily in polar solvents. What Does Isoelectric Point Tell You?Īt the isoelectric point, the molecule has no net charge, which means it is not attracted or repelled by charged particles in the environment. Amino acids with acidic side chains, such as aspartic acid and glutamic acid, have low isoelectric points, while those with basic side chains, such as arginine and histidine, have high isoelectric points. This value also wields the power to prognosticate the behavior of proteins under diverse conditions, envisioning scenarios involving chromatography or electrophoresis with remarkable accuracy.Įach amino acid has a different isoelectric point, which is influenced by the number and type of charged groups present in the molecule. The isoelectric point serves a significant purpose, serving as a metric to gauge a molecule's acidity or basicity, elements that profoundly influence its solubility, stability, and activity. However, at the magical isoelectric point, the amino acid stands in its glory as a zwitterion, gracefully adorned with both positive and negative charges. In contrast, at higher pH levels, the amino group stands proud as it sheds its proton (NH2), while the carboxyl group welcomes protonation (COOH), engendering a negative charge for the molecule. 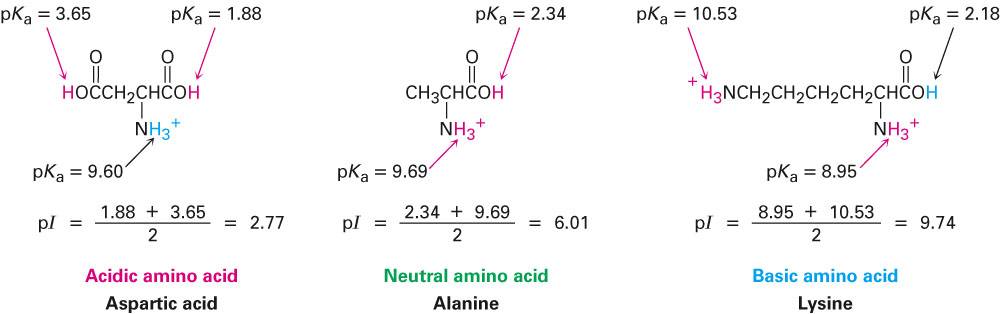
At lower pH levels, the amino group happily embraces protonation (NH3+), while the carboxyl group resists such a transformation (COO-), resulting in an overall positive charge for the molecule. These groups exhibit characteristics of weak acids and bases, respectively, and their protonation or deprotonation hinges on the pH of their surroundings. These amino acids possess a dual nature, with an amino group (-NH2) on one end and a carboxyl group (-COOH) on the other. To fully grasp the intricacies of this concept, it is paramount to acknowledge that amino acids form the fundamental building blocks of proteins. When we focus specifically on amino acids, this moment occurs precisely when the molecule's positive and negative charges are perfectly balanced, a delicate equilibrium dictated by the pKa values assigned to the amino and carboxyl groups. Picture this: no positive or negative charges prevail, as an equal count of these charged entities coexist harmoniously. Then, for individual proteins, pI predictions based on different pKa sets are presented alongside the molecular weight and amino acid composition.The isoelectric point, also referred to as the zwitterion's pH, signifies the pivotal moment when a molecule attains a neutral charge. The scatter plot (the virtual 2D-PAGE plot) with the predicted isoelectric points versus molecular weight for all proteins is presented at the top. Biology Direct 11:55.Įxemplary output of the IPC calculator for the Mycoplasma genitalium G37 proteome (476 proteins). Reference: Kozlowski LP (2016) IPC - Isoelectric Point Calculator. Peptide and protein datasets used in the study and the precalculated pI for the PDB and SwissProt databases are available for large-scale analysis and future development. Isoelectric point, the pH at which a particular molecule carries no net electrical charge, is an critical parameter for many analytical biochemistry and proteomics techniques, especially for 2D gel electrophoresis (2D-PAGE), capillary isoelectric focusing (cIEF), X-ray crystallography and liquid chromatography–mass spectrometry (LC-MS).Īccording to the benchmarks, IPC outperformed previous algorithms by at least 14.9% for proteins and 0.9% for peptides (on average, 22.1% and 59.6%, respectively), which corresponds to an average error of the pI estimation equal to 0.87 and 0.25 pH units for proteins and peptides, respectively. Output: pI values predicted by >15 methods alongside with virtual 2D-PAGE plot IPC – Isoelectric Point Calculator is a web service and a standalone program for the estimation of protein and peptide isoelectric point ( pI) using different sets of dissociation constant ( pKa) values, including two new, computationally optimized pKa sets. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
